CBHA
CBHA is a member of a recently synthesized family of hybrid polar compounds that have been shown to be inhibitors of HDAC and potent inducers of transformed cell growth arrest and terminal differentiation at micromolar (LD50 range, 1–4 μM) concentrations.
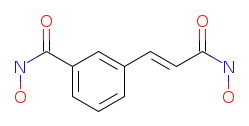
IUPAC Name: N-hydroxy-3-[(E)-3-(hydroxyamino)-3-oxoprop-1-enyl]benzamide
Molecular Weight: 222,20 g/mol
Molecular Formula: C10H10N2O4
SMILES: ONC(=O)c1cc(C=CC(=O)NO)ccc1
Canonical SMILES: C1=CC(=CC(=C1)C(=O)NO)C=CC(=O)NO
InChIKey Identifier: OYKBQNOPCSXWBL-SNAWJCMRSA-N
CAS Number: 174664-65-4
Melting point: n/a °C
Solubility: soluble in DMSO
2D Molfile: Get the molfile
Synonyms: m-Carboxycinnamic acid bis-Hydroxamide; M-carboxycinnamic acid bishydroxamide
This compound inhibits growth of human neuroblastoma xenografts in vivo alone and synergistically with retinoic acid.
References:
1) Dennis C. Coffey, Martha C. Kutko, et al. The Histone Deacetylase Inhibitor, CBHA, Inhibits Growth of Human Neuroblastoma Xenografts in Vivo, Alone and Synergistically with All-Trans Retinoic Acid. Cancer Research 61, 3591–3594, May 1, 2001.
2) Richard D. Glick, Steven L. Swendeman, et al. Hybrid Polar Histone Deacetylase Inhibitor Induces Apoptosis and CD95/CD95 Ligand Expression in Human Neuroblastoma. Cancer Research 59, 4392–4399, September 1, 1999.
3) Gipsy Majumdar. Maria Johnson, et al. Epigenetic regulation of cardiac muscle-specific genes in H9c2 cells by Interleukin-18 and histone deacetylase inhibitor m-carboxycinnamic acid bis-hydroxamide. Mol Cell Biochem (2008) 312:47–60.

|


