
 |
|
| Home | Vorinostat | CI-994 | MS-275 | BML-210 | M344 | NVP-LAQ824 | Panobinostat | Mocetinostat | PXD101 | MS-275MS-275 (SNDX-275; MS-27-275; Entinostat) preferentially inhibits HDAC 1 compared with HDAC 3 and has little or no effect against HDACs 6 and 8. The amine group of the benzanilide moiety might act as a hydrogen bond donor or could be involved in other electrostatic interactions, which are necessary for inhibitory activity. 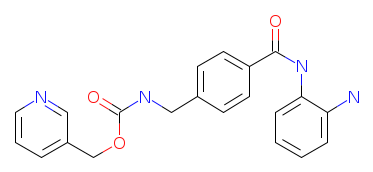
IUPAC Name: pyridin-3-ylmethyl 4-(2-aminophenylcarbamoyl)benzylcarbamate Currently, Entinostat (in combination with azacitidine) is in a clinical phase I/II trials for myelodysplastic syndromes, chronic myelomonocytic leukemia, and acute myeloid leukemia with multilineage dysplasia. This compound (in combination with azacitidine) is also in clinical phase I/II trials for patients with recurrent advanced nonsmall-cell lung cancer. References: |
| CBHA | PCI-24781 | ITF2357 | Valproic Acid | Trichostatin A | Sodium Butyrate | |
|
© 2010-2019 www.hdacis.com |